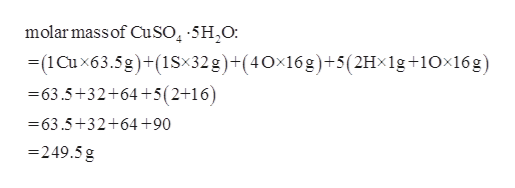 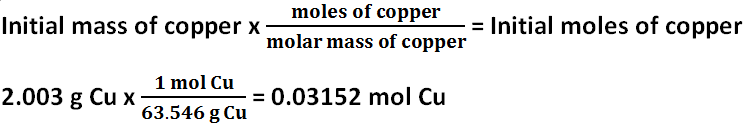
O (Oxygen) 4 × 15.9994 = 63.9976.Instead, we find evidence that Pb2 ions can directly exchange with Ba2 to create mixed (Ba,Pb)SO4 surfaces. Multiply the number of atoms by the atomic weight of each element found in steps 1 and 2 to get the mass of each element in SO4: Molar Mass (g/mol) S (Sulphur/Sulfur) 1 × 32.065 = 32.065. 182.09 g/mol bMolar Mass / Molecular Weight of (NH4)2SO4: Ammonium sulfate Wayne Breslyn 620K subscribers Subscribe 562 75K views 2 years ago Explanation of how to find the molar mass of (NH4)2SO4: Ammonium. 247.52 g/mol d Calculate the molar mass of C8H6O4. 28.12% e Calculate the molar mass for Mg (ClO4)2. Date: 05:36 AM Size: 190 KB License: applebees.skillport Calculate the mass percent composition of sulfur in Al2 (SO4)3. 
Show transcribed image text.Finally, we can use the formula above to calculate the molar mass of Cu: molar mass of Cu = mass / amount of substance = 0.36 g / 7.761 x 10^-3 mol = 46.35 g/mol Therefore, the …Download windows update troubleshooter windows 10 - Windows Update Troubleshooter Author: Microsoft Corp. Note: Atomic mass and …Question: Calculate the formula mass of sodium sulfate heptahydrate, Na, SO4.7H20, in atomic mass units (amu or u). As we know that the atomic mass is equal to the mass of one mole of compound, the atomic mass of \ is 96.0676 \. If we write this as a calculation, it looks like this: (1 atom x 23 grams/mole Na) (1 atom x 35.5 grams/mole Cl) = 58.5 grams/mole NaCl 2. So4 mass If we have a chemical compound like NaCl, the molar mass will be equal to the molar mass of one atom of sodium plus the molar mass of one atom of chlorine.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
